Electroless Plating
 Plating is a technique that has been done for hundreds of years, and is still relevant for modern technology today. It is used for many purposes including decorating objects, corrosion inhibition, hardening, friction reduction, to improve paint adhesion, alter conductivity, for radiation shielding and other purposes.
Plating is a technique that has been done for hundreds of years, and is still relevant for modern technology today. It is used for many purposes including decorating objects, corrosion inhibition, hardening, friction reduction, to improve paint adhesion, alter conductivity, for radiation shielding and other purposes.
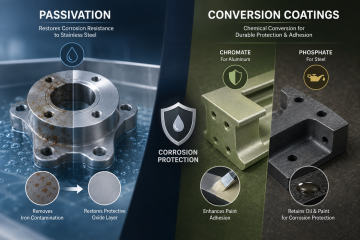
There are many types of plating including electroless plating. Electroless plating is also known as chemical or auto-catalytic plating. Electroless plating is a technique of plating metal by chemical rather than electrical means. Electroless plating is beneficial because it allows the coating of metals and plastics by a process of controlled autocatalytic, or self-continuing, reduction. It is a non-galvanic plating method involving simultaneous reactions which occur without the use of external electrical power. When hydrogen is released by a separate electroless solution-derived compound called the reducing agent (usually sodium hypophosphite or formaldehyde) it causes a chemical reaction to occur. It oxidizes, producing a negative charge on the surface of the part.
An electroless solution typically consists of a source of metal ions, a reducing agent, a complexant for the metal ions to keep these dissolved ions in solution, usually a pH adjustment buffer, sometimes a few ppm of a catalytic poison to stop unwanted plating on the walls fo the tank, and oxygen gas dissolved in solution.
Electroless plating has seen a major expansion in the area of plastics. Many consumer goods are coated by the electroless method because it creates durable and attractive surfaces. Recently it has been used extensively for metalizing printed wiring boards. Because electroless plating allows a constant metal ion concentration to bathe all parts of the object, it deposits metal evenly along edges, inside holes, and over irregularly shaped objects which can otherwise be difficult to plate. Electroless plating is also sometimes used to deposit a conductive surface on a nonconductive object which allows it to then be electroplated.